Combined Holter Records Both ECG, Blood Pressure
4.5 (583) · € 38.99 · En Stock
April 13, 2010 – The U.S. Food and Drug Administration (FDA) recently cleared a combined electrocardiogram (ECG) Holter and ambulatory blood pressure monitoring recorder and software analysis system. The Vasomedical-Biox Model 2301 system is among the first combined systems to simultaneously and continuously record and store ECG and blood pressure data for 24 hours. The ability to view these parameters simultaneously, side by side gives the physician more useful information to evaluate the patient's cardiovascular status. In addition to being a compact device that replaces two separate recorders, it also provides interactive blood pressure recording at times of certain cardiac abnormalities. This can enhance the diagnostic value of the recorded data, while increasing patient comfort and ease of use. For more information: www.vasomedical.com

Portable Ultrasound ECG Holter Monitor Blood Pressure Monitor - China ECG Holter Monitor, Holter ECG
SH-EP Combined ECG Holter and ABP System from Farum : Get Quote, RFQ, Price or Buy
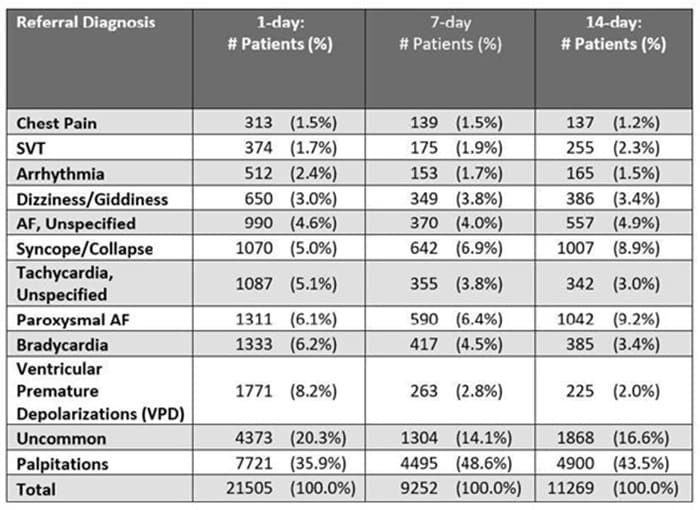
New ambulatory monitoring data at ESC - News
Important tips for sleeping with a Holter monitor
Veroval® 2 in 1 ECG and Blood Pressure Monitor

Holter ECG Ultimate holter ecg monitor - Norav Medical

FDA TLC5000 12 lead 24Hour Holter ECG/EKG System Monitor Analyzer PC Software - International Society of Hypertension

Dynamic 24hours 12 Lead ECG/EKG Holter Monitor Alalyzer Software CONTEC TLC5000 - International Society of Hypertension
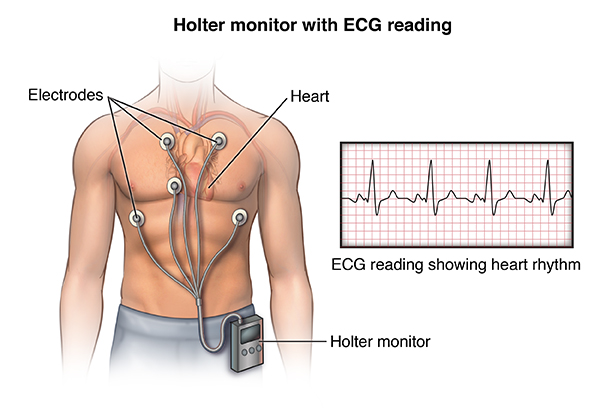
Holter Monitor Johns Hopkins Medicine