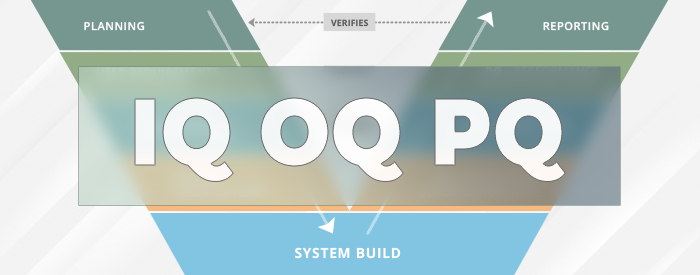
What is IQ OQ PQ in Software Validation?
4.6 (100) · € 20.99 · En Stock
If you work in a regulated industry (FDA), you know about IQ, OQ, PQ testing—let CSols Inc. de mystify these scripts on your behalf.

FDA Guidance on Software Validation: Terminology
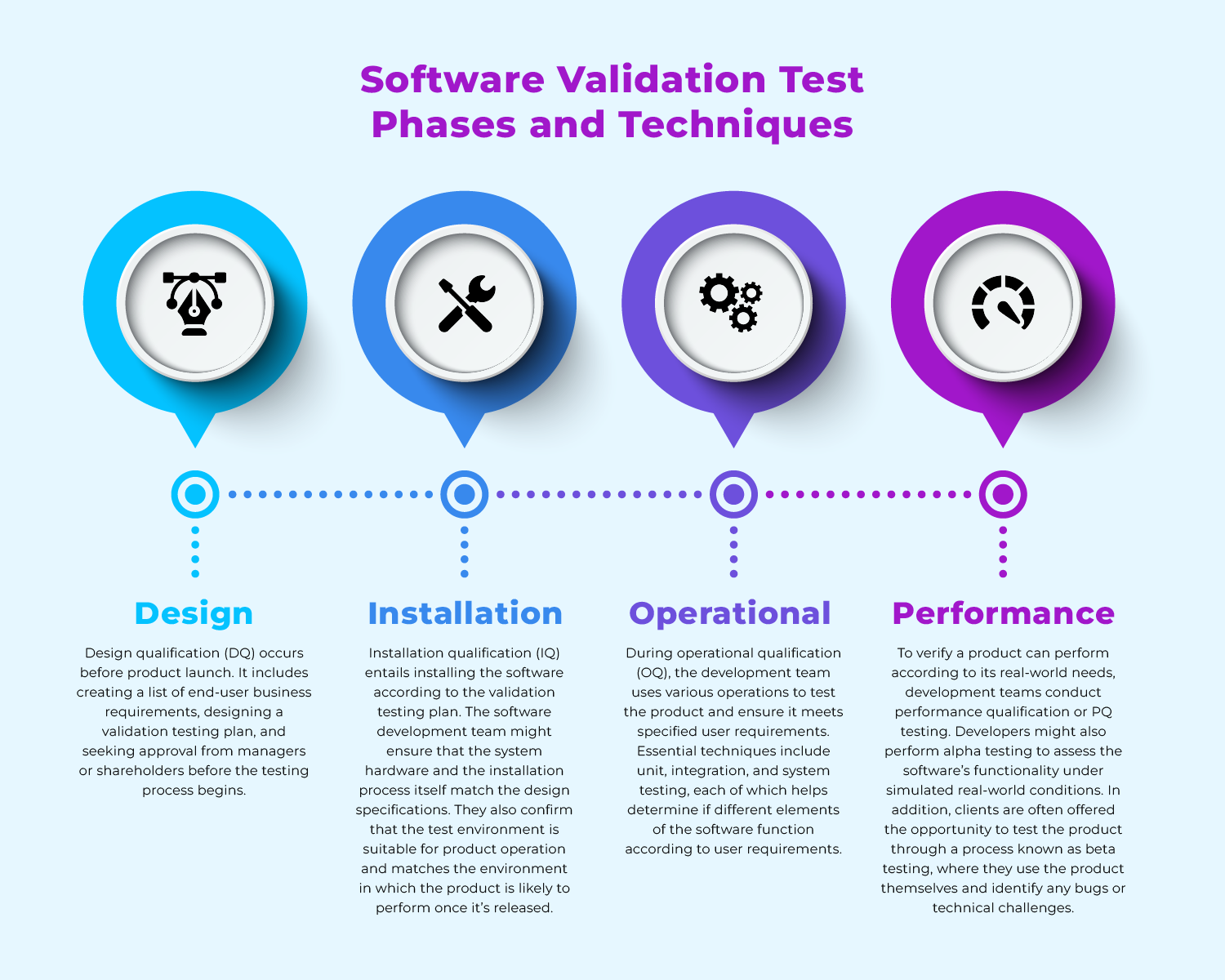
What Are IQ OQ PQ, The 3 Q's Of Software Validation Process
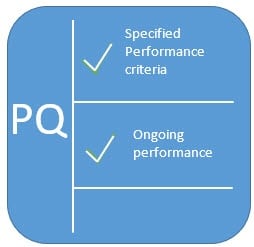
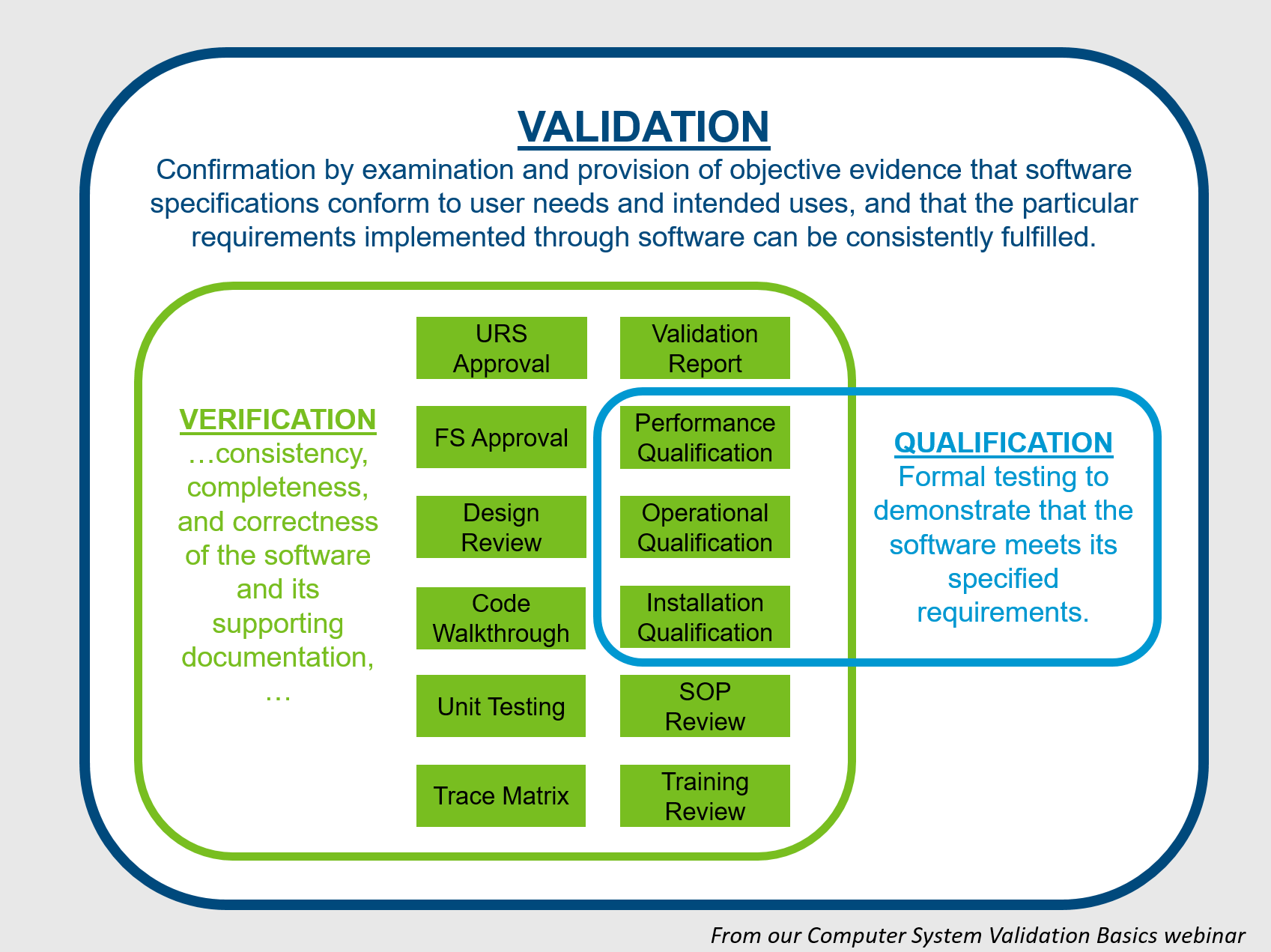
The OQ/PQ Validation Protocol is used to document the system's ability to meet the functional requirements specifications (OQ) and user requirements

CSV Template - Validation Protocol - OQ or PQ
Qualification, IQ OQ PQ, Validation Services

What Are IQ OQ PQ: The 3 Q's Of Software Validation Process
What Are IQ OQ PQ, The 3 Q's Of Software Validation Process
How Validation Testing Can Enhance Your Customer Satisfaction
What is Computer System Validation and How Do You Do It?
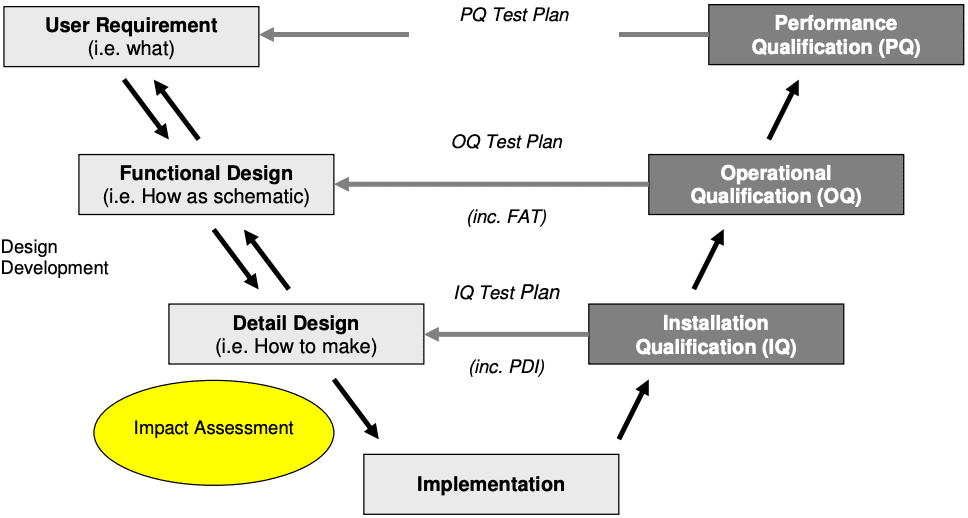
What are IQ OQ PQ? Why are they critical to the Pharma Industry?

iq oq pq in pharmaceuticals for software or equipment process validation training

Why you don't need an IQ, OQ or PQ for your validated system audit
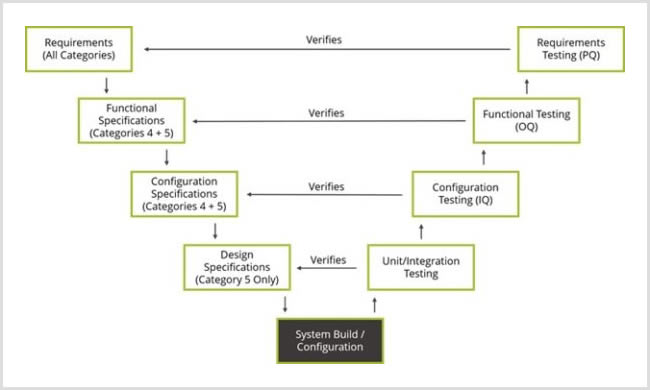
What You Should Know About CSV in Pharma

What Are IQ OQ PQ: The 3 Q's Of Software Validation Process