Classification of Bases - on Basis of Solutibility, Ionising Capacity
4.8 (339) · € 24.00 · En Stock
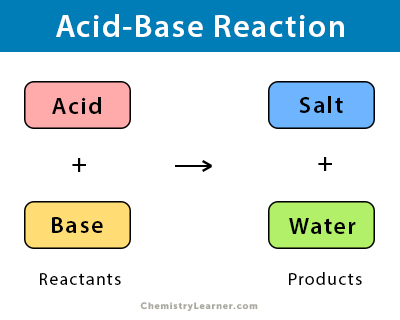
Bases can be categorised based on different properties:On the Basis of Solubility:Bases can be classified asSolubleandInsoluble.Soluble Bases: These are bases which are dissolvable in water. These are also calledAlkalis. For Example : Sodium Hydroxide (NaOH), Potassium Hydroxide (KOH)Insoluble Bases

Common Ion Effect - Statement, Explanation, and Examples

Disproportionation of Pharmaceutical Salts: pHmax and Phase-Solubility/pH Variance
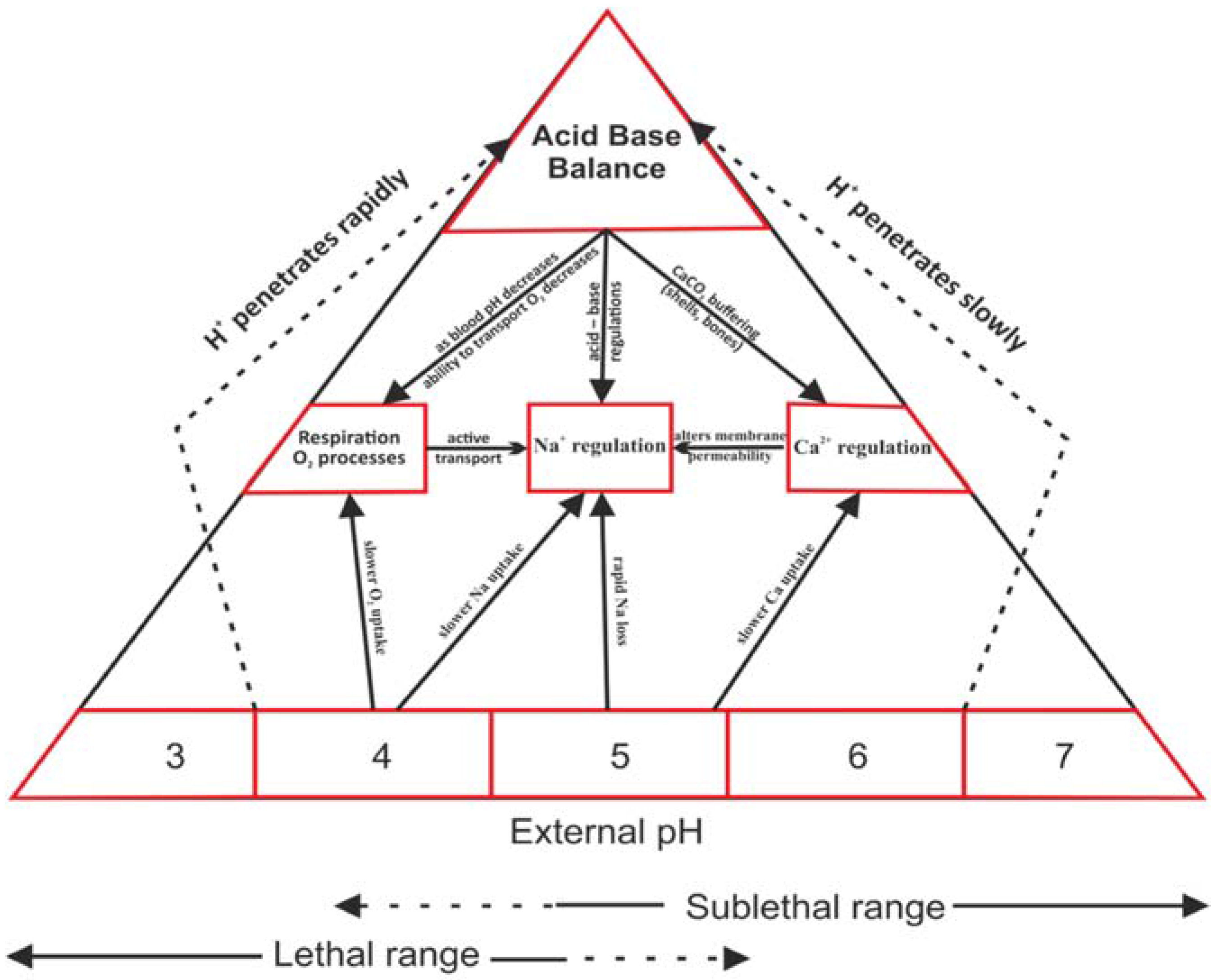
Water, Free Full-Text
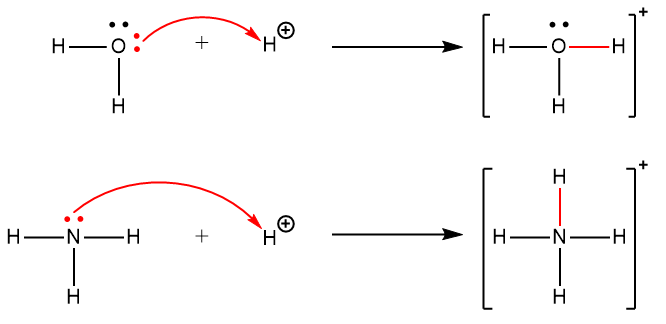
5.6 – Lewis Acids & Bases – General Chemistry for Gee-Gees

Relative Strengths of Acids and Bases - LabXchange

Fasted and fed state human duodenal fluids: Characterization, drug solubility, and comparison to simulated fluids and with human bioavailability - ScienceDirect

Phase Behavior and Crystallization Kinetics of a Poorly Water-Soluble Weakly Basic Drug as a Function of Supersaturation and Media Composition

Degree of Ionization - an overview
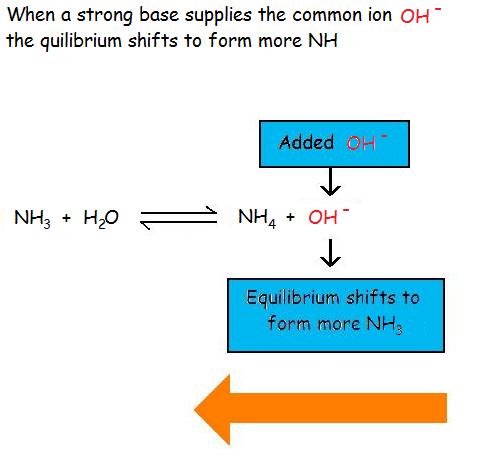
7.3: Common-Ion Effect in Solubility Equilibria - Chemistry LibreTexts